 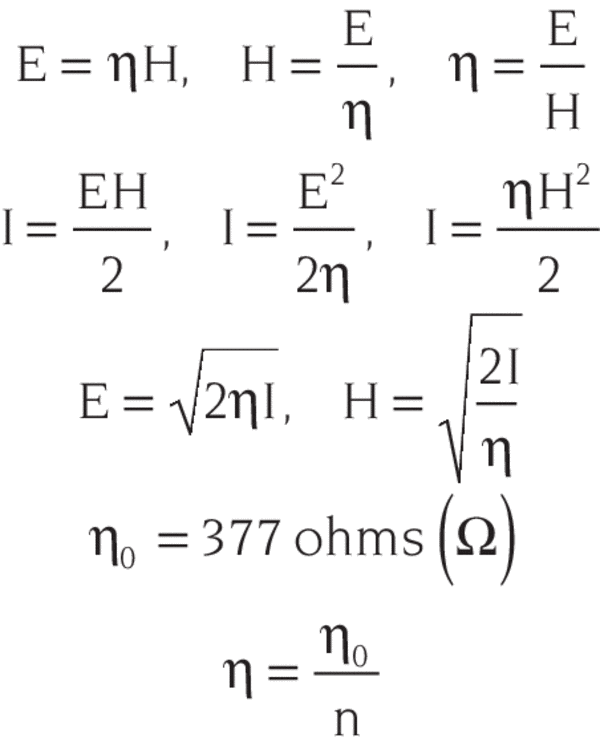
In 1729, Pierre Bouguer discovered this law initially. Very often many scientists are given credits for the discovery of this law which are as follows: We can determine the concentration of various substances in cell structures by measuring their absorbing spectra in the cell. We use Beer Lambert Law to conduct a qualitative and quantitative analysis of biological and dosimetric materials that may contain organic or inorganic materials. The same method can be used in determining the molar absorbance of bilirubin in blood plasma samples. In electromagnetic spectroscopy, we use electromagnetic radiation (we may take UV rays), which scans the tablet and determines the qualitative (drug present) and the quantitative (concentration) property of the tablet. Though we may know the drug, then the question arises about what its molar concentration is. Let’s suppose we have a tablet and we don’t know which drug is present in it. To analyze the drugs, for that, let’s take an example of a tablet: Now, let’s understand the applications of Beer Lambert law: The important use of the Beer Lambert law is found in electromagnetic spectroscopy. We know the value of b, thus we can determine the value of ε easily. Thus, the slope of the graph is given by εb. It is observed that a straight line passing through the origin is obtained. This value is determined by plotting the values of absorbance (on the y-axis) against various values of concentrations (on the x-axis) and determining the slope of the line. The graphical method is used to determine the value of the Molar Extinction Coefficient (ε). 
When monochromatic radiation passes through a homogeneous medium, then the rate of decrease in the intensity of the transmitted radiation with the increase in the thickness of the medium and the concentration of the solution varies directly with the intensity of incident radiation. So, the final Beer Lambert law statement is: Ε = Molar absorption coefficient or molar absorptivity in m -1 cm -1 = k’ x k’’ Now, we get our simplified expression as:įor determining the Beer Lambert law equation, let’s combine equation (2) & (4), and take the log of these, we get:Įquation (6) is the required Beer Lambert Law Formula. This expression says that the absorbance of light in a homogenous material/medium is directly proportional to the concentration of the sample. 
We can express this statement mathematically as When monochromatic light passes through a ‘transparent medium’, the rate of decrease of transmitted radiation with the increase in the concentration of the medium is directly proportional to the intensity of the incident light. This expression says that the absorbance of light in a homogenous material/medium is directly proportional to the thickness of the material/medium. We can write the equation (1) in the following way as well: Here, I t = Intensity of transmitted radiation So, the absorbance \= k’I o (Here, the negative sign indicates the decrease in the intensity of the transmitted radiations).(1)Įquation (1) says that the rate of decrease in the intensity to the thickness is directly proportional to the incident radiation. The intensity of transmitted radiation = I t 
Here, the intensity of incident radiation = I o, and Now, passing electromagnetic radiation (incident radiation or we may use UV rays) to the drug sample, some of the light may get absorbed, and the rest of it gets transmitted. Let’s say, we have a clear sample of a drug with a polished surface around its container. It relates the weakening of the intensity of the light to the characteristics of the medium through which it is travelling. Beer Lambert law is one of the popular topics in analytical chemistry. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
